
Land plants — plants that live primarily in terrestrial habitats and form vegetation on earth — are anchored to the ground through their roots, and their performance depends on both the belowground soil conditions and the aboveground climate. Plants utilize sunlight to grow through the process of photosynthesis where light energy is converted to chemical energy in chloroplasts, the powerhouses of plant cells. Therefore, the amount and quality of light perceived by chloroplasts through light absorbing pigments, such as chlorophyll, is a defining factor in plant growth and health. A substantial amount of the chemical compounds produced during the conversion of light energy to chemical energy, termed photoassimilates (mainly sugars), is translocated to the plant root compartment and invested in the surrounding soil to sustain microbial growth.
Consequently, roots harbour complex microbial communities of bacteria and filamentous eukaryotes (i.e., fungi and oomycetes), and the composition of these communities profoundly influences plant performance. However, the extent to which plants can take advantage of belowground microbes to orchestrate aboveground stress responses remains largely unexplored. Now, in a new study published in Nature Plants, Stéphane Hacquard and his colleagues from the Department of Plant-Microbe Interactions at the MPIPZ in Cologne, Germany, shed light on these aboveground-belowground connections.
To tackle this question, the first author of the study Shiji Hou performed experiments where the aboveground light conditions and the belowground microbial conditions could be controlled. By comparing the growth of Arabidopsis thaliana (Thale Cress) grown in the absence of root microbes (i.e., germ-free) to those colonized by a complex community of 183 bacteria, 24 fungi and 7 oomycetes, the researchers observed that the presence of microbes rescued the plant growth-deficiency observed under low light conditions. Inoculation experiments with leaf pathogens further indicated that plants colonized by microbes were also more resistant to aboveground leaf pathogens than germ-free control plants, indicating that the presence of root microbes can promote both plant growth and defense under low light.
By comparing growth and defense responses of colonized plants between the two light conditions, the scientists observed that investment in growth under low light conditions came at the cost of defense, since microbiota-induced defense responses were reduced and plants were more susceptible to leaf pathogens under low light. Based on this observation, the authors of the study then hypothesized that when light conditions are suboptimal, plants favor microbe-induced growth over microbe-induced defense responses. To test this hypothesis, the researchers screened different A. thaliana mutants to identify those that failed to invest in growth under low light. Consistent with their hypothesis, the identified mutants were better at resisting leaf pathogens instead. Furthermore, the scientists found that the presence of the host transcription factor MYC2 was crucial to tip the balance in favor of microbiota-induced growth instead of microbiota-induced defense under low light conditions.
The researchers then went on to investigate whether the make-up of the microbial community belowground could explain aboveground investment in growth at the expense of defense under low light. To do this, they analyzed the composition of the root microbiota across the different A. thaliana mutants and observed that the bacterial community composition was markedly different depending on whether the different plants invested in growth under low light. This experiment led to the identification of 67 bacterial strains that were predicted to be associated with plant growth rescue under low light. To test a potential causal link, the researchers prepared three different bacterial communities composed of either: 1) all 183 strains, 2) the 183 strains lacking the 67 strains predicted to be important for growth rescue or 3) the 67 strains alone. Remarkably, A. thaliana wild-type plants colonized with the 67-member community invested in growth under low light, whereas those colonized by the community lacking these bacterial strains did not, instead favoring better resistance to leaf infection by pathogens.
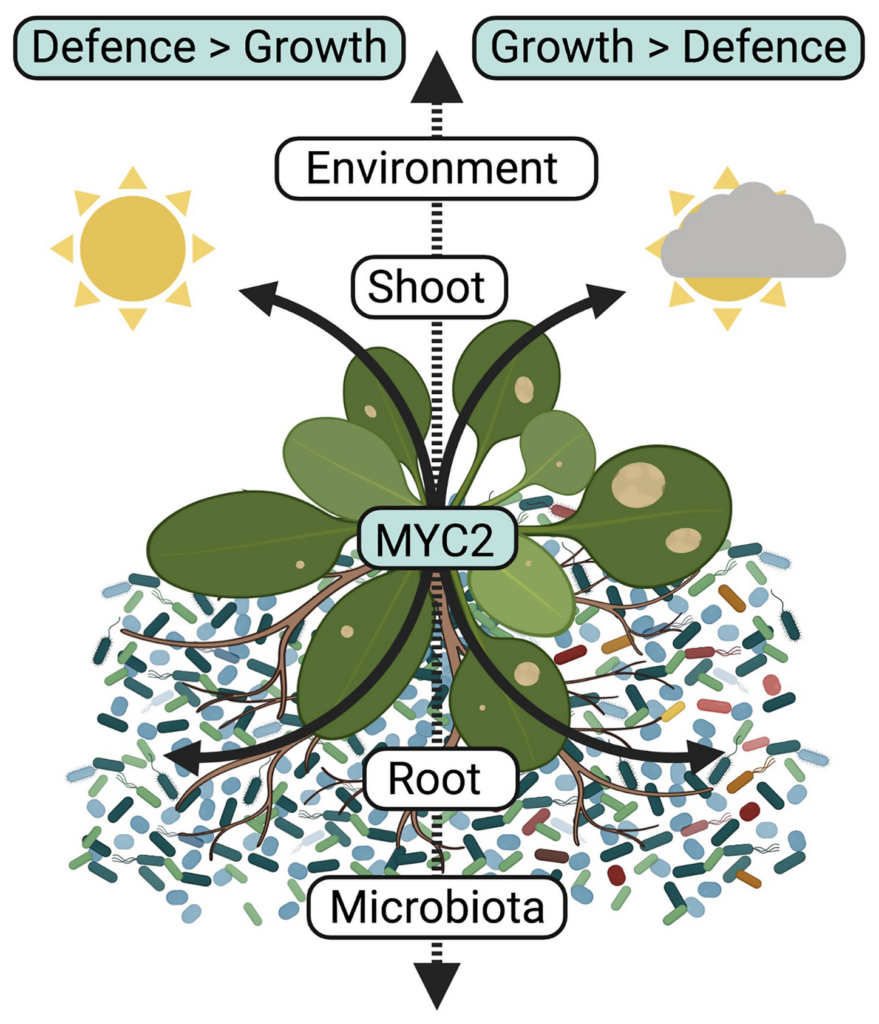
In the words of study lead Stéphane Hacquard: “Our results suggest that plant growth and defense responses are engaged in different feedback loops with the root microbiota depending on aboveground light conditions. It is likely that light-induced change in root exudation profiles is an important mechanism that stimulates the growth of particular beneficial bacterial root commensals that boost plant growth, in the expense of defense responses under low light.” The observation that microbiota-root-shoot-circuits exist in plants is reminiscent of recent results obtained in the context of the microbiota-gut-brain axis in animals, where a direct link between gut commensals and brain functions was uncovered. The results suggest that bacterial root and gut commensals have important functions in modulating stress responses not only locally, but also in distant host organs.
These findings have important applications for utilizing belowground microbes to promote aboveground stress responses in plants. By applying the knowledge gained in this study it would now be conceivable to design synthetic microbial communities with modular functions that could be used to promote plant resistance to particular biotic or abiotic stresses, and ultimately promote plant health in nature.
 Neem Oil: Your Natural Shield for Thriving Indoor Plants – A Complete Guide to Pest Protection
Neem Oil: Your Natural Shield for Thriving Indoor Plants – A Complete Guide to Pest Protection 10 Nutrient-Dense Organic Foods to Boost Your Energy Without Caffeine
10 Nutrient-Dense Organic Foods to Boost Your Energy Without Caffeine Rye is healthy, thanks to an interplay of microbes
Rye is healthy, thanks to an interplay of microbes Precise reason for health benefits of dark chocolate: Thank hungry gut microbes
Precise reason for health benefits of dark chocolate: Thank hungry gut microbes